|
Purification was then effected by multiple recrystallization steps to give a yield of about 320 mg per kilogram of dried roots.ĭue to our need for significant quantities of cyclopamine, we required a practical large-scale isolation scheme. After storing the eluent at 5☌ for several months, a mixture of alkaloids crystallized. In previous isolations, cyclopamine was extracted by allowing room-temperature benzene to percolate through dried, ground roots and rhizomes of Veratrum californicum which had been wetted with 5% ammonia. 
We also provide unambiguous assignments of the carbon and proton resonances by using the multinuclear spectra and the spin coupling networks. In this paper we report the rapid isolation and purification of cyclopamine with excellent yields. Thus, in order to obtain the large amounts of compound necessary for our animal research we required a significantly more efficient method to extract and purify cyclopamine from the roots and rhizomes of Veratrum californicum. The isolation and purification procedures specifically designed for Veratrum alkaloids that have been reported previously were also lengthy and of relatively low yield. However, because this would in aggregate be a nontrivial complex multistep synthesis of very low net yield, and because the concentration of cyclopamine in Veratrum californicum is so high, vide infra, we opted to isolate the compound from this natural source. 
Second, the Wolff-Kishner reduction of jervine has been reported to yield cyclopamine. First, the synthesis of 17-acetyl-5α-etiojerva-12,14,16-trien-3β-ol from simple chemicals has been reported by Johnson et al, and Masamune et al reported its conversion into the cyclopamine relative jervine, where the carbon-11 methylene in cyclopamine is oxidized to ketone in jervine. However, because it is very expensive commercially (~$10/mg), the large quantities required for research in animals, and especially in large mammals as required in our own cardiac research, would at present be prohibitively expensive.Ī totally synthetic approach to obtaining cyclopamine can be envisioned. As one important example, cyclopamine has been shown to be an extremely effective inhibitor of the growth and survival of specific tumors in vitro and in vivo, such that cyclopamine, cyclopamine analogs, and related alkaloids have the potential to become important cancer chemotherapeutic agents. Cyclopamine is thus a very useful reagent for probing and modifying the activity of the important and widely studied Hh pathway. We also provide unambiguous assignments of the carbon and proton resonances by using the multinuclear spectra and the spin coupling networks.ConclusionThis method could meet a very real need within diverse scientific communities by allowing cyclopamine to become more readily available.Ĭyclopamine is a naturally occurring steroidal alkaloid teratogen that interferes with the Hedgehog (Hh) signaling pathway by binding to Smoothened, a heptahelical transmembrane receptor that is a critical Hh response element. (the Corn Lily or Western false hellebore). To use cyclopamine as a tool to explore and/or inhibit the Hedgehog pathway in vivo, a substantial quantity is required, and as a practical matter cyclopamine has been effectively unavailable for usage in animals larger than mice.ResultsIn this paper, we report a rapid and efficient isolation and purification of large quantities of cyclopamine from the roots and rhizomes of Veratrum californicum Dur. 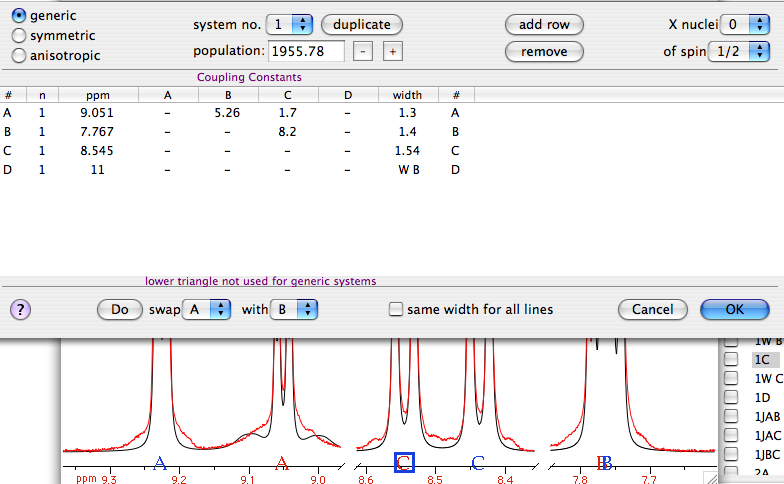
Cyclopamine, a naturally occurring steroidal alkaloid, specifically inhibits the Hedgehog pathway by binding directly to Smoothened, an important Hedgehog response element. However, it may induce malignancies in a number of tissues when constitutively activated, and it may also have a role in other forms of normal and maladaptive growth. BackgroundThe Hedgehog signaling pathway is essential for embryogenesis and for tissue homeostasis in the adult.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
